by
John R. Fischer, Senior Reporter | February 25, 2019
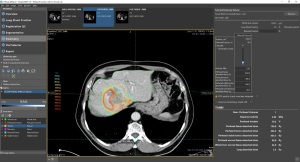
Simplicit90Y facilitates the design of
more personalized planning for
radioembolization treatments
The FDA has given thumbs-up to Mirada Medical for the launch of its Simplicit90Y dosimetry software across the U.S.
Designed in collaboration with BTG for personalized treatment of patients with liver cancer, the solution speeds up dosimetry planning and improves workflow of Y90 TransArterial Radioembolization, a form of treatment in which doctors use a catheter to deliver tiny glass spheres of Yttrium-90 (Y90) radiation directly into cancer cells of the liver where they become permanently lodged and continued to deliver radiation to the tumor over the course of several weeks.
"TheraSphere Y90 glass microspheres are specifically engineered to deliver high doses of radiation to liver tumors while sparing normal tissue," Mirada Medical told HCB News. "The result is a powerful, targeted and well-tolerated therapy that may lead to patients becoming eligible for curative therapies."



Ad Statistics
Times Displayed: 365785
Times Visited: 7088 Quality remanufactured Certified Centrifuges at Great prices! Fully warranted and backed by a company you can trust! Call or click for a free quote today! www.Centrifugestore.com 800-457-7576
Performing a TheraSphere procedure requires merging data sets from multiple types of imaging modalities to develop a treatment plan and calculate the appropriate dose of TheraSphere. Simplicit90Y does this through its digital processing, review and reporting of medical images, creating a streamlined, single interface in which users can incorporate multimodal images, and use automated and semi-automated tools for segmentation, manipulate data and expedite analysis.
It also offers data display, quality control, image manipulation and quantification analysis, all of which help to standardize and create reproducible dosimetry planning for more consistent and personalized treatment based on an individual patient's needs, as well as post-treatment validation for the assessment of the absorbed dose delivered to the patient.
"The software should be of interest to providers looking to optimize radioembolization therapy," said Mirada Medical. "It allows them to offer patients a personalized treatment, but can also help improve the efficiency and consistency of treatment."
A recent study found radioembolization to be an effective option for
treating children with hard-to-reach liver cancer that is resistant to chemotherapy and cannot be surgically removed to help improve survival time.
In the U.S., TheraSphere is approved under a Humanitarian Device Exemption (HDE) for use in radiation treatment or as a neoadjuvant to surgery or transplantation in patients with unresectable hepatocellular carcinoma (HCC) who can have placement of appropriately positioned hepatic arterial catheters. The device is also indicated for HCC patients with partial or branch portal vein thrombosis/occlusion, when clinical evaluation warrants the treatment. The effectiveness of this device for this use has not been demonstrated.
The system is cleared for use in Canada and is CE marked. It is approved under a Humanitarian Device Exemption in the U.S. for use in radiation treatment; as a neoadjuvant to surgery or transplants in patients with unresectable hepatocellular carcinoma (HCC), who are approved for placement of appropriately positioned hepatic arterial catheters; and for HCC patients with partial or branch portal vein thrombosis, when warranted by clinical evaluation. The effectiveness of the device for this last use has not been demonstrated.
BTG plans to showcase it from March 23-28 at the Society of Interventional Radiology (SIR) congress in Austin.

