by
John R. Fischer, Senior Reporter | January 10, 2019
The FDA has given Quantib the OK to begin distribution of its radiology software, Quantib ND, in the U.S.
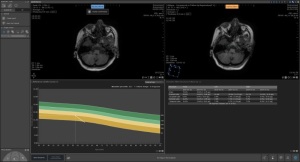
Designed to support MR brain scan reading, the solution measures brain atrophy and alerts radiologists to the presence of white matter hyperintensities, changes in the brain that signal the onset of aging, dementia and multiple sclerosis.
"There are many ways for AI to add value for radiologists," Arthur Post Uiterweer, CEO of Quantib, told HCB News. "Quantib ND offers very detailed, and most importantly, quantified information for cases where this information is currently not obtained as it is too time consuming to derive manually from the scans. Additionally, referring physicians such as neurologists and geriatricians are increasingly asking for volume measurements that can assist in assessing whether a brain is aging normally or not. Quantib ND will provide more standardized output and it eliminates interobserver variance."



Ad Statistics
Times Displayed: 173774
Times Visited: 3176 For those who need to move fast and expand clinical capabilities -- and would love new equipment -- the uCT 550 Advance offers a new fully configured 80-slice CT in up to 2 weeks with routine maintenance and parts and Software Upgrades for Life™ included.
Extensively tested and proven to be reproducible, the software is composed of fully automatic segmentation of lobes and hippocampus for objective assessments of atrophy development, as well as white matter hyperintensity segmentation for simple monitoring of neurological changes that take place in patients with dementia and MS. It also can compare the brain volume of patients to a vast database of thousands of unbiased subjects using reference centile curves, derived from the population-based Rotterdam Scan Study.
Another component is its follow-up feature for tracking neurodegeneration over time. This creates an accurate alignment of images at subsequent time points that enables accurate detection and staging of atrophy, combined with WMH tracking. It is further strengthened by the software’s ability to visualize new and previously identified lesions using color-coded overlays.
In addition, radiologists can use the solution’s editing feature to fully control the final results before sending them to the PACS, with patient data protected due to the installation of Quantib ND taking place on the premises of the provider where patient scans are processed behind firewalls.
The company previously
gained FDA and CE authorization in August 2016 for its software, Quantib Brain, which automatically extracts biomarkers from brain MR scans that strongly correlate with the onset and development of neurodegenerative and inflammatory diseases such as Alzheimer's and multiple sclerosis.
Brain is a plug in to GE's Advantage Workstation, making it only available to those with GE IT systems. Quantib ND, in contrast, is vendor neutral and can be used on any PACS system. Radiologists of any background are capable of using the tool whether they carry a neuro subspecialization or not.
"Our roadmap builds on our capabilities to apply machine learning to complex 3D MR images for organ segmentation and tissue classification. Future products will also focus on products that fit in our vision of democratizing radiology. In prostate cancer, for example, we see how our prototype algorithms could help radiologists in the future to become much better at detecting cancer on multiparametric MR scans," said Uiterweer. "It is the vision of Quantib to democratize radiology, meaning we want to enable any radiologist, urban or rural, to perform important tasks that currently are only viable for specialized centers."